Pantoprazole for Injection IP
Price 56.5 INR/ Piece
MOQ : 100 Pieces
Pantoprazole for Injection IP Specification
- Assay
- Not less than 98% (on dried basis)
- CAS No
- 138786-67-1
- Appearance
- White to off-white lyophilized powder
- Molecular Formula
- C16H15F2N3O4S
- Storage
- Store below 25C, protected from light
- Indication
- Treatment of gastroesophageal reflux disease (GERD), peptic ulcer, and Zollinger-Ellison syndrome
- Origin of Medicine
- India
- Dosage Form
- Lyophilized powder for injection
- Expiration Date
- 24 months from manufacturing date
- Pacakaging (Quantity Per Box)
- 1 vial
- Salt Composition
- Pantoprazole Sodium
- Usage
- Proton pump inhibitor for gastric acid suppression
- Dosage
- 40 mg/vial
- Medicine Type
- INJECTION
- Grade
- Pharmaceutical Grade
- Solvent Provided
- Sterile Water for Injection
- pH Range
- 8.5 to 10.5
- Packing Type
- Vial with flip-off seal
- Route of Administration
- Intravenous
- Reconstitution Instructions
- Dissolve content with 10 mL Sterile Water for Injection
- Country of Export
- India
- Therapeutic Class
- Antiulcer agent
- Shelf Life
- 2 years
- Prescription Required
- Yes
- Contraindications
- Known hypersensitivity to pantoprazole or any component
About Pantoprazole for Injection IP
- ACTIVE INGREDIENT
- PANTOPRAZOLE SODIUMM EQUIVALENT TO 40 MG PANTOPRAZOLE.
- ALSO CONTAINS EDETATE DISODIUM(OFTEN~1 MG PER VIAL) A A STABILIZER)
- PHYSICAL FORM
- LYOPHILIZED (FREEZE-DRIED) POWDER IN A SINGLE-DOSE VIAL.
- COLOR: WHITE TO OFF-WHITE CAKE/POWDER.
Effective Acid Suppression for GI Conditions
Pantoprazole for Injection IP is specially formulated to provide rapid suppression of gastric acid secretion, making it highly effective in the treatment of conditions such as GERD, peptic ulcers, and Zollinger-Ellison syndrome. Its pharmaceutical-grade quality ensures reliable therapeutic outcomes.
Easy Reconstitution and Administration
This medication comes as a white to off-white lyophilized powder in a vial, with Sterile Water for Injection provided as a solvent. Healthcare professionals can easily reconstitute the powder with 10 mL of sterile water, allowing for precise intravenous dosing and faster patient relief.
Trusted Export Quality from India
Manufactured and supplied from India, Pantoprazole for Injection IP meets international pharmaceutical standards. Each batch undergoes stringent quality checks, with an assay of not less than 98% on a dried basis, ensuring consistent efficacy and safety for global patients.
FAQ's of Pantoprazole for Injection IP:
Q: How should Pantoprazole for Injection IP be reconstituted and administered?
A: Pantoprazole for Injection IP should be dissolved in 10 mL of Sterile Water for Injection. Once reconstituted, it is administered intravenously as directed by a healthcare professional.Q: What are the primary medical uses of this injection?
A: This injection is used for the treatment of gastroesophageal reflux disease (GERD), peptic ulcers, and Zollinger-Ellison syndrome, helping reduce gastric acid secretion.Q: When is this medication typically prescribed by a doctor?
A: Pantoprazole for Injection IP is usually prescribed when oral administration is not suitable or when rapid acid suppression is required, always under a doctor's supervision.Q: Where should the reconstituted injection be stored before use?
A: The injection should be used immediately after reconstitution. The unopened vial should be stored below 25C and kept protected from light.Q: What precautions should be taken before using this medication?
A: Patients with known hypersensitivity to pantoprazole or any component of the formulation should avoid using this medication. It should always be administered under medical supervision.Q: What are the key benefits of choosing this intravenous formulation?
A: This formulation provides fast and reliable acid suppression, is suitable for patients unable to take oral medications, and meets pharmaceutical-grade standards for safety and efficacy.Q: Is a prescription required to obtain Pantoprazole for Injection IP?
A: Yes, this injection is a prescription-only medication and should be used only under the guidance of a qualified healthcare provider.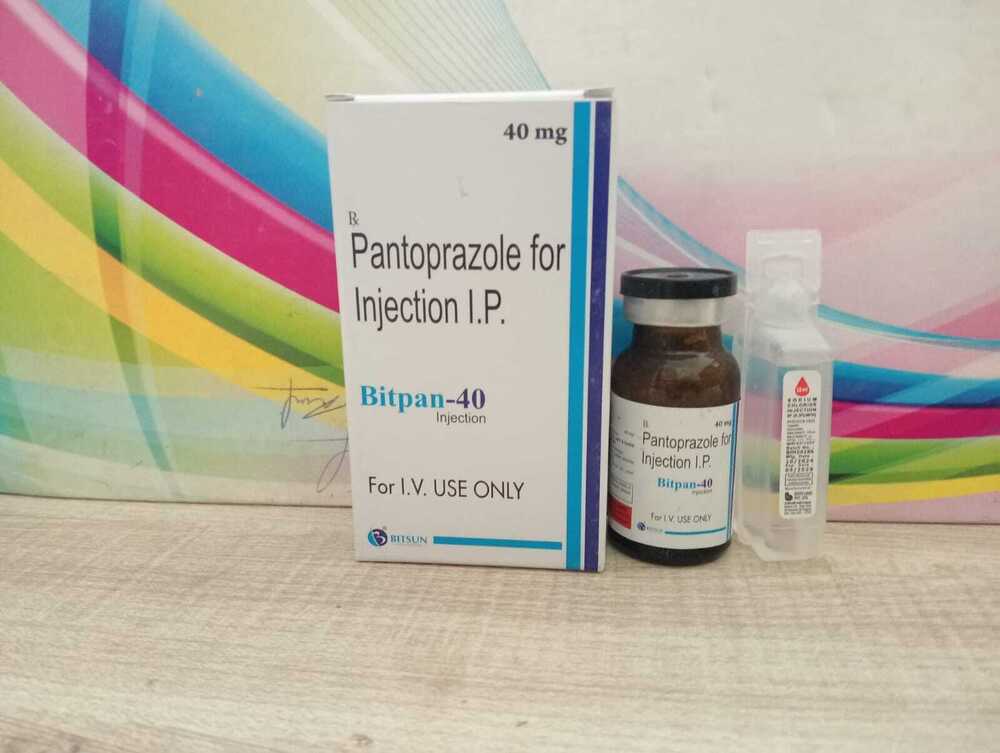
Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
More Products in Pharmarceutical Injection Category
1 Gm Cefoperazone And Sulbactam Injection
Price 125 INR / Pack
Minimum Order Quantity : 500 Packs
Storage : Cool Place
Origin of Medicine : India
Dosage Form : Injection
Grade : Pharmaceutical Grade
Cefoperazone And Sulbactam Injection
Price 160 INR / Pack
Minimum Order Quantity : 500 Packs
Storage : Cool Place
Origin of Medicine : Imported
Dosage Form : Injection
Grade : Medical
Cinric-S 1 Injection
Price 215.0 INR / Piece
Minimum Order Quantity : 500 Pieces
Storage : Dry Place
Origin of Medicine : India
Dosage Form : Injection
Grade : medical

 Send Inquiry
Send Inquiry


 Send Inquiry
Send Inquiry Send SMS
Send SMS